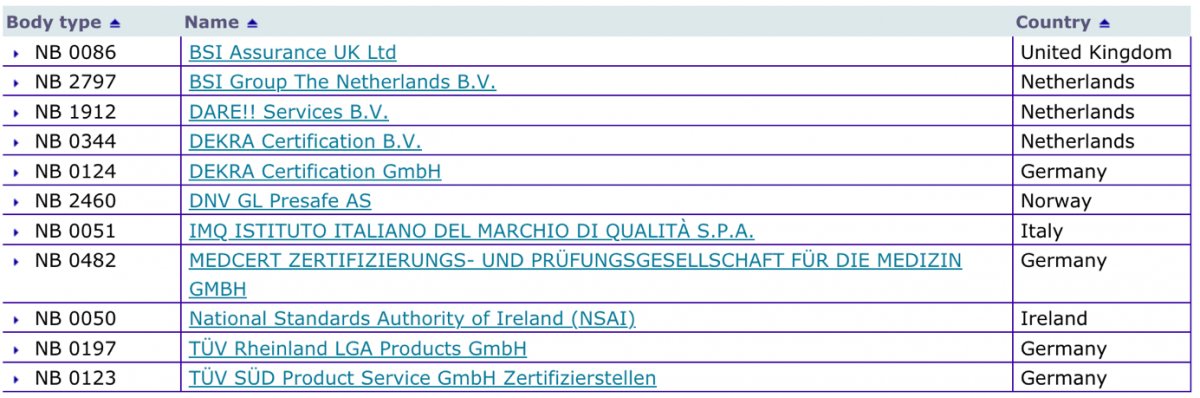
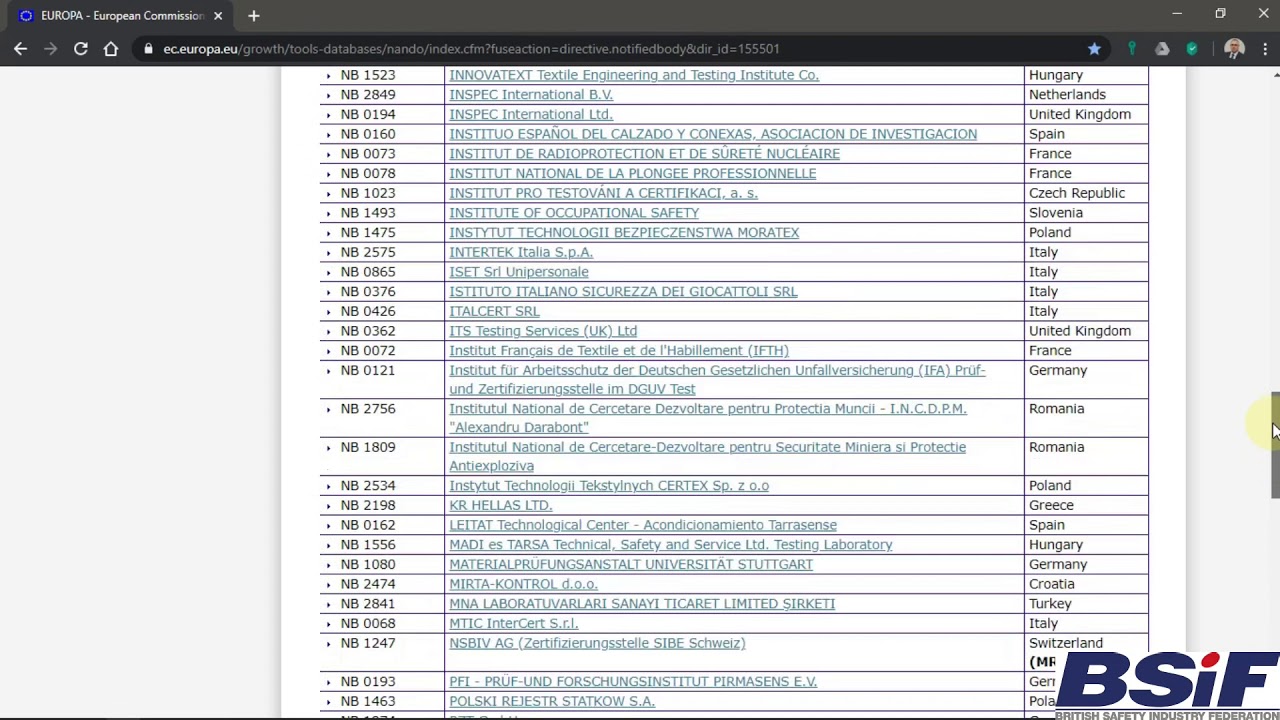
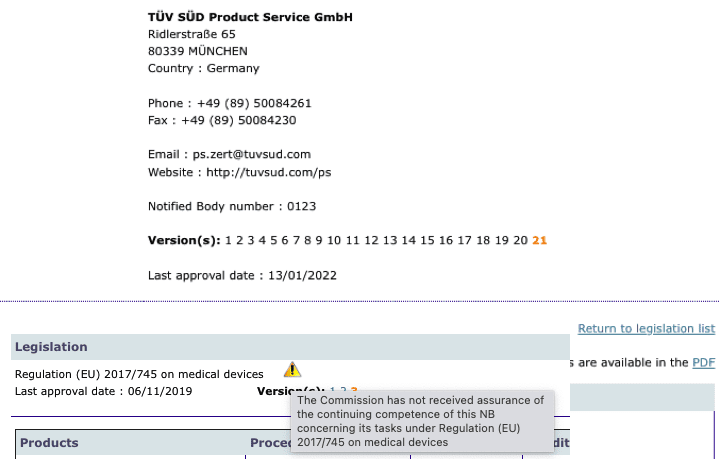
MDR: 26 Notified Bodies on NANDO & Swiss economic operator's requirements updated! · MDlaw – Information platform on European medical device regulations
Informational - New designated Notified Body for Regulation (EU) 2017/745 ( MDR) included in Nando – IMQ ISTITUTO ITALIANO DEL MARCHIO DI QUALITÀ S.P.A.
![Glossary] EU MDR and IVDR: The core terms you should know - Pharma Serialization, Aggregation and Track and Trace Software by SoftGroup Glossary] EU MDR and IVDR: The core terms you should know - Pharma Serialization, Aggregation and Track and Trace Software by SoftGroup](https://www.softgroup.eu/wp-content/uploads/2020/10/Untitled-design-38-1280x720.png)
Glossary] EU MDR and IVDR: The core terms you should know - Pharma Serialization, Aggregation and Track and Trace Software by SoftGroup






![Xplood]081 | by ɳɑɳɖɵ ɑɾɾʉɗɑ Nando Arruda © All rights rese… | Flickr Xplood]081 | by ɳɑɳɖɵ ɑɾɾʉɗɑ Nando Arruda © All rights rese… | Flickr](https://live.staticflickr.com/4023/4701505689_851b495793_b.jpg)




![BSI News] BSI Netherlands Notified Body (NB 2797)도 MDR 심사 기관으로 지정되었습니다! | BSI Blog BSI News] BSI Netherlands Notified Body (NB 2797)도 MDR 심사 기관으로 지정되었습니다! | BSI Blog](http://bsiblog.co.kr/wp-content/uploads/BSI-Netherland-MDR.png)