
Calculate the mass percentage of carbon, hydrogen and oxygen in ethanol - CBSE Class 11 Chemistry - Learn CBSE Forum

Section 4 - Mass Percent Composition | Math Tutor DVD - Online Math Help, Math Homework Help, Math Problems, Math Practice!

SOLVED: Acetic acid (HC2H3O2) is the active ingredient in vinegar. Calculate the mass percent composition of oxygen in acetic acid.

Mass Percent of a Solution Made Easy: How to Calculate Mass % or Make a Specific Concentration - YouTube



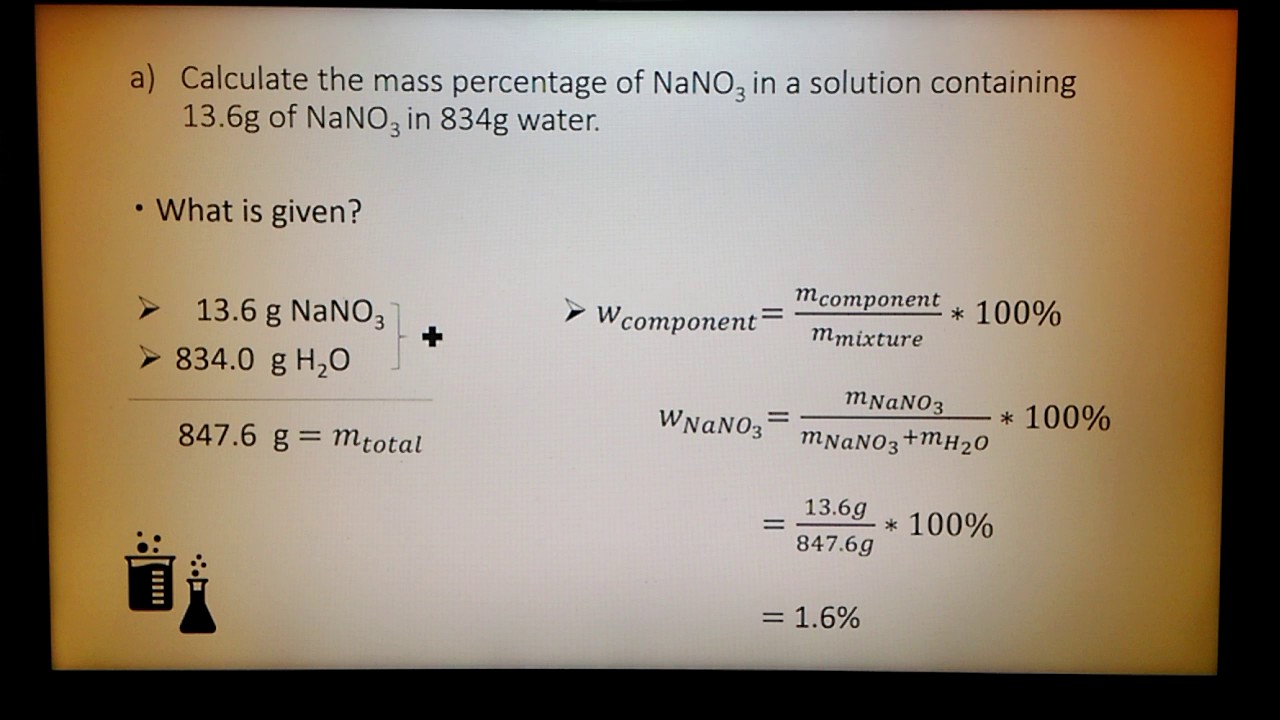






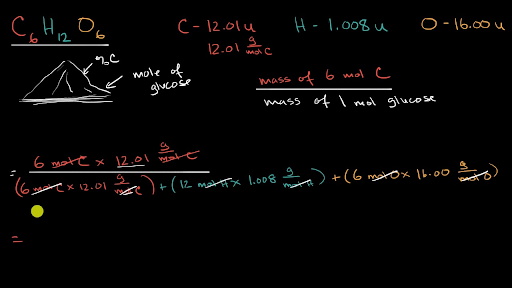







:max_bytes(150000):strip_icc()/mass-percent-composition-example-609567_V2-01-89c18a9d30ea43b494d09b81f7ffefc1.png)
:max_bytes(150000):strip_icc()/mass-percent-composition-example-609567_V2-01-89c18a9d30ea43b494d09b81f7ffefc1.png)