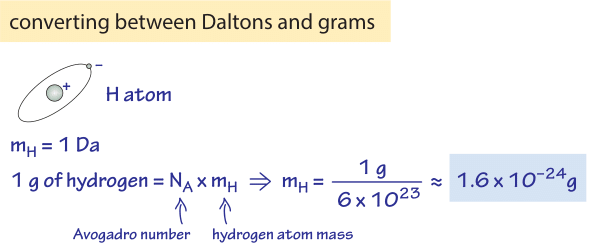AQUEOUS SOLUTIONS Solute Concentration Molecular Weight = Sum of weight of all atoms in a molecule (expressed in Daltons). For example: Determine a mole. - ppt download

Dalton used the percentages of elements in compounds and the chemical formulas to deduce the relative masses of atoms Unit is the amu(atomic mass. - ppt download