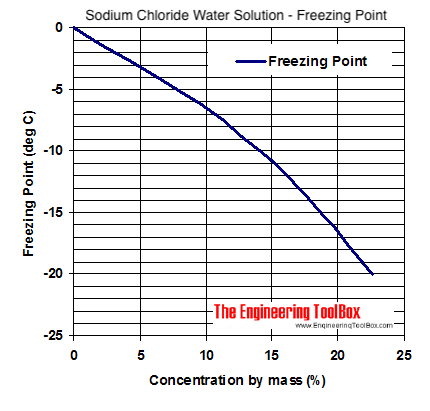
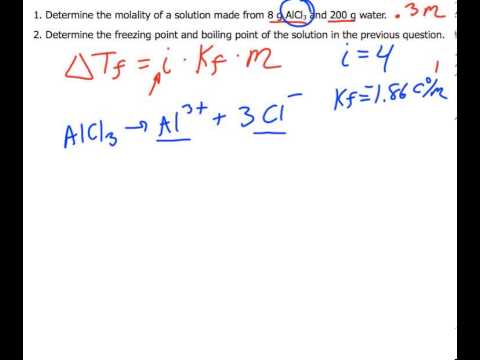SOLVED: Determine the freezing point of 1.80 g of MgSO4 (FW 120.09 g/mol) dissolved in 1L of solution. Assume i=2.0 for MgSO4. The molal freezing point depression constant of water is 1.86

How to calculate the freezing point of an equimolar ethanol: water mixture using colligative properties - Chemistry Stack Exchange

Calculate the freezing point depression and boiling point elevation of a solution of `10.0 g` of... - YouTube
Calculate the freezing point of a solution containing 60 g of glucose (Molar mass = 180 g mol^-1) in 250 g of water. - Sarthaks eConnect | Largest Online Education Community
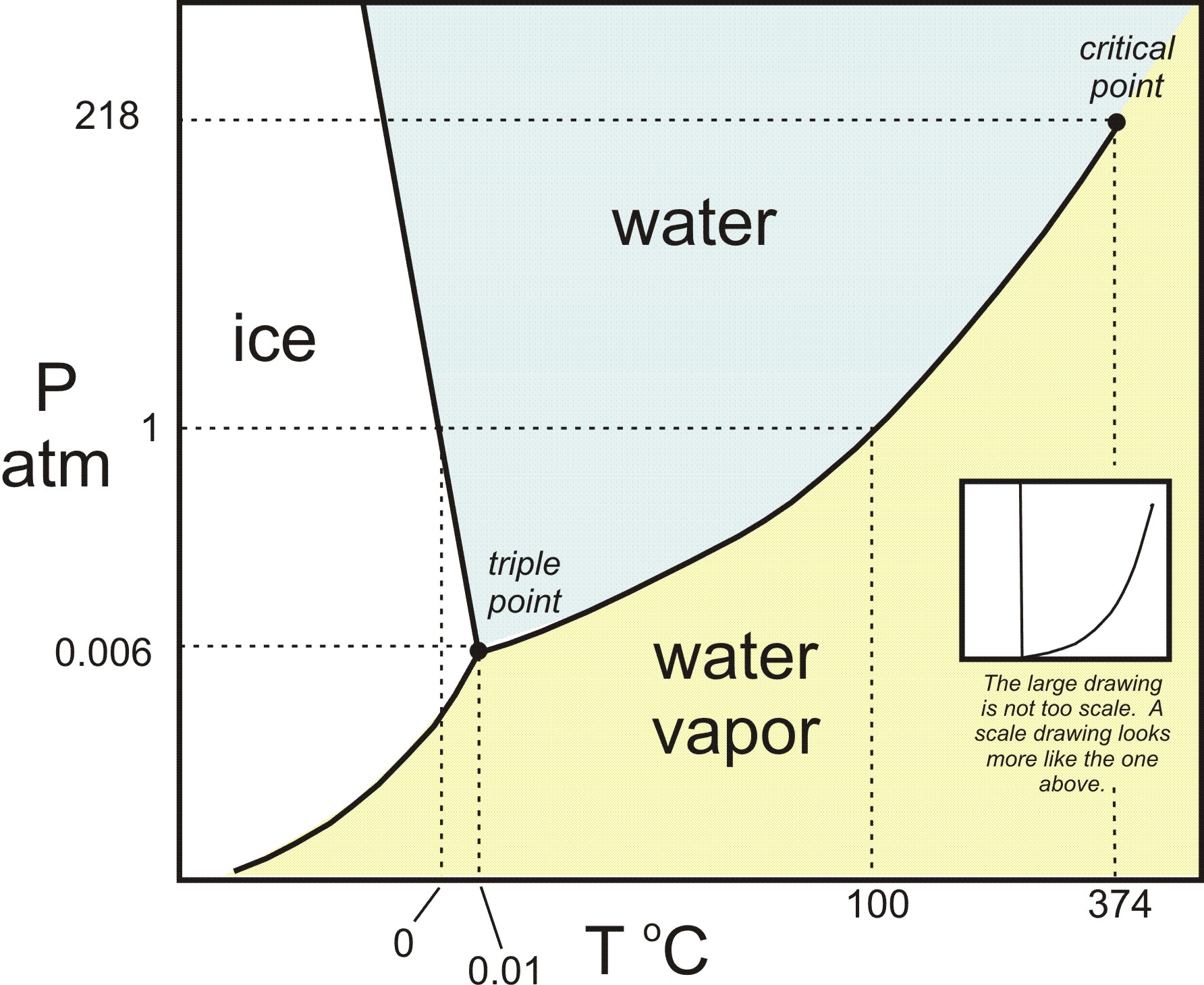
Calculate the change in freezing point for ice if the pressure changes from "1 atm" to "3 atm"? | Socratic

Calculate the freezing point and the boiling point at 1 atmosphere of a solution containing 30 g cane sugar (molecular mass 342 ) and 150 g water.Given : Kb = 0.513 and Kf = 1.86
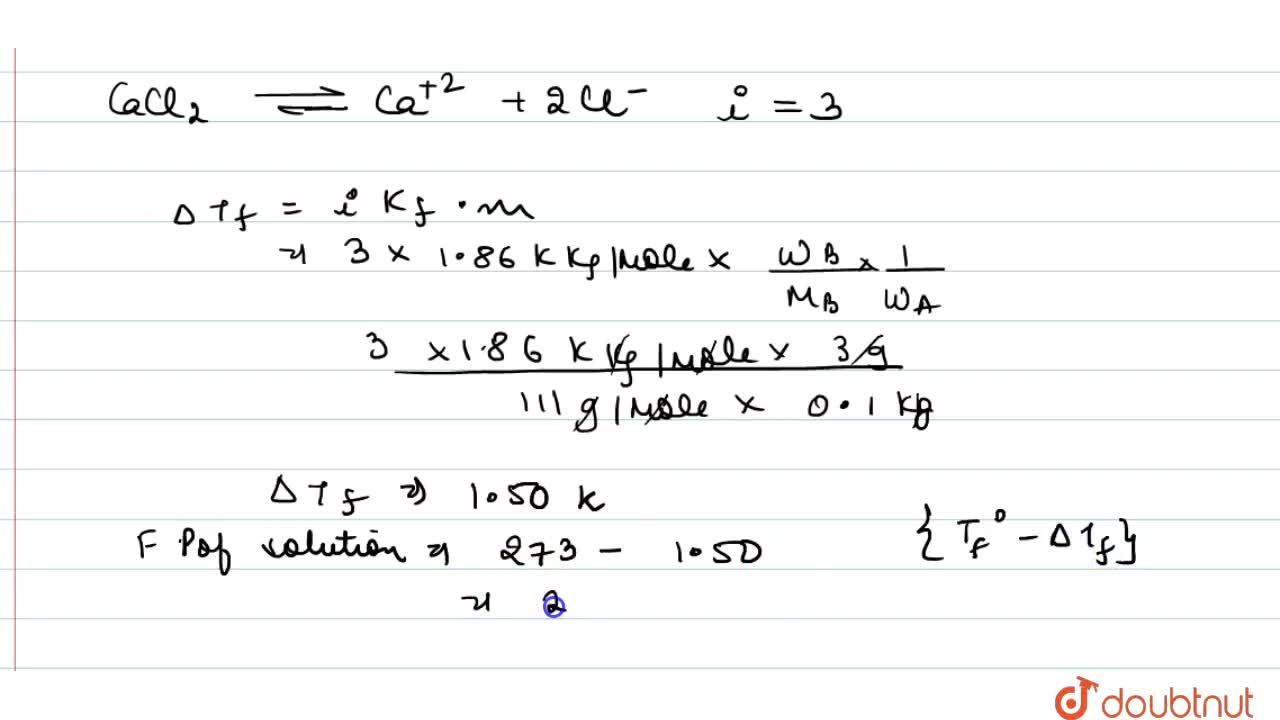
Calculate the freezing point of a solution when 3 g of CaCI(2) (M=111 g mol^(-1)) was dissolved in 100g of water assuming that CaCI(2) undergoes complete ionisation (K(f) "for water"=1.86 K kg

A solution of urea in water has a boiling point of 100.18 ^o C. Calculate the freezing point of the solution. (K f for water is 1.86 K kg mol ^-1 and

A brief introduction to freezing point depression THE COLLIGATIVE PROPERTIES OF MOLALITY AND FREEZING POINT DEPRESSION grownextgen.org. - ppt download