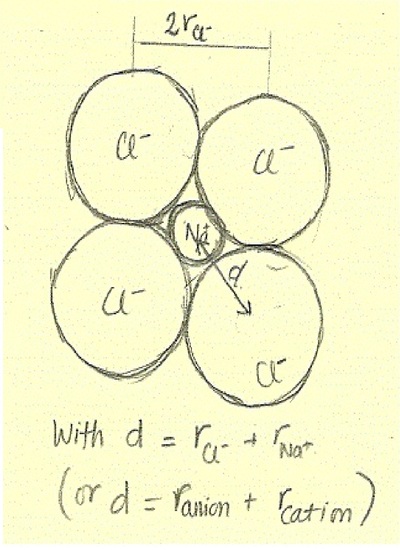Niobium has a density of 8.57 g/cm3 and crystallizes with the body-centered cubic unit cell. Calculate the radius of a niobium atom - Chemistry Stack Exchange
What is the formula to calculate the radius of an orbit of the atom and velocity of the specific shell of the atom.

The value of covalent bonf length of Si - C is 1.93 A . Covalent radius of carbon atom is 0.77 A . Calculate the covalent radius of silicon atom.

Atomic radius and ionic radius of F(g) and F(g)^- are 72 and 136 pm prespectivley. Calculate the ratio and percentage increase in terms of volume during formation of F(g)^(-) form F(g) .
Welcome to Chem Zipper.com......: How to calculate percentage (%) ionic character in covalent compounds?

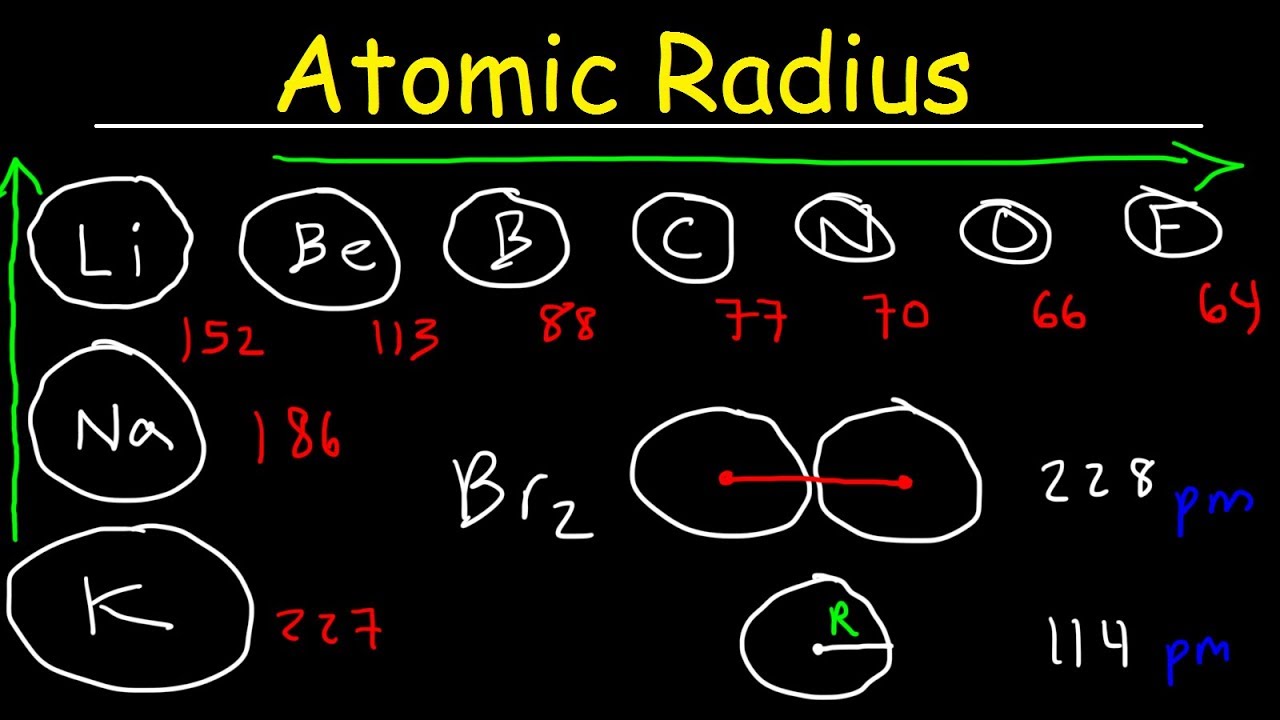
What do you mean by covalent radius? from Chemistry Classification of Elements and Periodicity in Properties Class 11 Karnataka Board